December 28, 2024
Aluminum alloy anodizing technology development
Aluminum alloy material has a small relative density, can be welded easily, is easy to form and process, and has higher mechanical strength. These characteristics make it widely used in various fields, especially in the aviation industry, where its application is increasing. Aluminum has a strong affinity with oxygen in the air. Even in dry air, it reacts easily with oxygen, forming a thin layer of non-porous amorphous Al2O3 on the surface. However, the naturally occurring oxide film is thin and has poor wear and corrosion resistance. This thin film does not meet the requirements for reliable protection.
To improve the corrosion resistance of aluminum alloy, surface treatment is necessary. Anodic oxidation is the most commonly used surface treatment for aluminum and aluminum alloys. The anodic oxide film formed on aluminum has good mechanical properties, corrosion resistance, friction resistance, and strong adsorption on the surface.
In aviation, aluminum alloy is the main material used in aircraft fuselage structures. When designing aircraft, the chosen structural materials must have high specific strength and specific stiffness to reduce the structural mass or increase economic efficiency. These materials should also have good processability. The skin, beams, ribs, trusses, spacer frames, and landing gear of aircraft are often made from aluminum alloy. Aluminum alloys are classified into nine categories based on different alloying elements, as shown in Table 1.
Table 1: Aluminum and Aluminum Alloy Grade Designations.
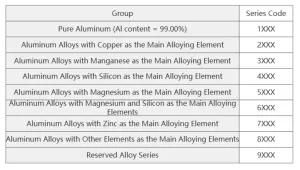
Among these, 7XXX and 2XXX aluminum alloys are the most widely used. The 7075 aluminum alloy offers high strength, good mechanical properties, and excellent corrosion resistance. The 2024 aluminum alloy is a heat-treated AI-Cu-Mg system alloy. It has high strength and good machining properties, making it suitable for critical aircraft components like the skin, skeleton, ribs, and beams.

To overcome the limitations of aluminum alloy surface performance expand its range of applications and extend its service life, surface treatment technology is essential. The anodic oxidation of aluminum alloys provides a surface with excellent corrosion resistance, good adhesion for organic coatings, high hardness, and wear resistance. It also creates an insulating film with high resistance. Aluminum alloy anodic oxidation technology meets various needs, offering excellent qualities such as corrosion resistance, wear resistance, decoration, adhesion, and functionality.
1. Aluminum Alloy Anodizing Technology
Aluminum alloy anodic oxidation is a dynamic equilibrium process between the growth and dissolution of the oxide film. The goal during anodic oxidation is to ensure that the growth rate of the oxide film exceeds the dissolution rate of the surface layer. There are several types of anodic oxidation technology: direct current anodic oxidation, alternating current anodic oxidation, and pulse current anodic oxidation. The resulting films can be classified into ordinary films, hard films, porcelain films, bright modification layers, and semiconductor barrier layers. Based on the electrolyte, anodizing can use sulfuric acid, chromic acid, oxalic acid, or mixed acid. Natural coloring anodic oxidation is also a common method.
1.1 Anodizing in Sulfuric Acid Solution
Ordinary sulfuric acid anodic oxidation produces a 0.5–20.0μm film with good adsorption, suitable for general protection or as a bonding base layer for paint coatings (e.g., for aircraft outer skins). The sulfuric acid anodic oxidation film is porous, with a void rate of around 35%. It has strong adsorption capabilities, making it easy to dye and widely used for decorative purposes. While the film offers good corrosion resistance, the fatigue performance of the base material can be affected, making it unsuitable for spot welding or riveted assemblies. It also retains parts of the electrolyte.
1.2 Anodizing with Chromic Acid Solution
The chromic acid oxide film is dense and tree-like in structure. After oxidation, it does not require a closed treatment. It has good adhesion with coatings, and its corrosion resistance is superior to that of the sulfuric acid oxide film, which is not closed. However, alloys with more than 4% copper content are generally unsuitable for chromic acid anodic oxidation. This method is more expensive and environmentally polluting compared to sulfuric acid anodic oxidation. Even with environmental protection measures, it increases the process cost, which limits its widespread use.
1.3 Boric Acid-Sulfuric Acid Solution Anodizing
Boric acid-sulfuric acid anodizing combines the benefits of chromic acid anodizing, including good covering ability, low concentration of tank solution, no C(Ⅶ), convenient solution treatment, and low environmental pollution. This method is known as an “environmentally friendly” surface treatment. The boric acid-sulfuric acid anodized film is thin and has low stress, unlike sulfuric acid anodizing, which is prone to cracks. Like chromic acid anodizing, the oxide film is highly elastic and has a dense structure.

1.4 Oxalic Acid Solution Anodizing
Oxalic acid anodizing produces an oxide film with a thickness of 8-20μm. This method, which was widely used in Japan and Germany, results in a thicker film with higher hardness and better wear and corrosion resistance. It also provides good electrical insulation and protective properties. However, oxalic acid anodizing is prone to electric breakdown and ablation, resulting in low qualification rates. It is also sensitive to chlorine ions, requiring higher applied voltages and energy consumption. Therefore, production costs are 3-5 times higher than with sulfuric acid anodizing.
1.5 Phosphoric Acid Solution Anodizing
Phosphoric acid anodizing was first researched and adopted by Boeing in the U.S. It is an environmentally friendly, low-toxicity, and low-cost anodizing method. The film formed is relatively thin and has large pores, making it suitable for filling with lubricants or other functional materials. However, due to its thinness, its application is somewhat limited compared to sulfuric acid and oxalic acid anodizing.
1.6 Mixed Acid Anodizing
Mixed acid anodizing uses a combination of sulfuric acid, oxalic acid, and other acids. This method improves the temperature range and oxidation efficiency and enhances the hardness and wear resistance of the oxide film. However, the complex electrolyte composition and higher cost compared to sulfuric acid anodizing limit its application.
2. Aluminum Alloy Anodic Oxidation Technology Research Status
In practical production, the pretreatment process, anodic oxidation, and film closure treatments sometimes fail to meet the required corrosion resistance standards. This can directly affect production efficiency. Researchers have focused on improving the pretreatment process, anodic oxidation, and film closure treatments to overcome these challenges.
2.1 Pre-treatment Process
The purpose of pretreatment is to remove oxides and oils from the surface and prepare a clean, flat, and activated surface for anodic oxidation. The process includes degreasing, alkali etching, light washing, and water washing. Alkali etching is the most critical and challenging step, as it removes oxides and activates the aluminum alloy surface. The temperature and time must be carefully controlled to avoid uneven corrosion or excessive corrosion.
2.2 Anodizing Process
Aluminum alloy anodic oxidation forms an oxidized film under the influence of an applied current in the electrolyte. The factors affecting the anodic oxide film’s performance include the electrolyte, additives, power supply type, and process conditions. Sulfuric acid anodizing is cost-effective, simple, and widely applicable, but it has limitations in low-temperature environments and high energy consumption. Chromic acid anodizing provides a thinner film with good corrosion resistance and paint adhesion, but its environmental impact is a concern.

2.3 Membrane Layer Sealing Treatment
Common sealing methods include hot water, steam, dichromate, inorganic salts, and organic compounds. These methods face challenges like high toxicity, high energy consumption, and low efficiency. Research into non-polluting, stable, and low-energy sealing processes, such as microwave sealing, nickel-free medium-temperature sealing, and rare earth salt sealing, is ongoing. Experts have investigated new sealing methods that enhance the corrosion resistance of aluminum alloys and improve sealing efficiency.
3. Conclusion
Anodic oxidation technology plays a crucial role in the production of aluminum alloy parts used in aircraft. As modern industries develop and environmental consciousness grows, the role of aluminum and its alloys in daily production and life is increasing. As a result, the anodic oxidation process must meet higher demands, with more diverse functional requirements.